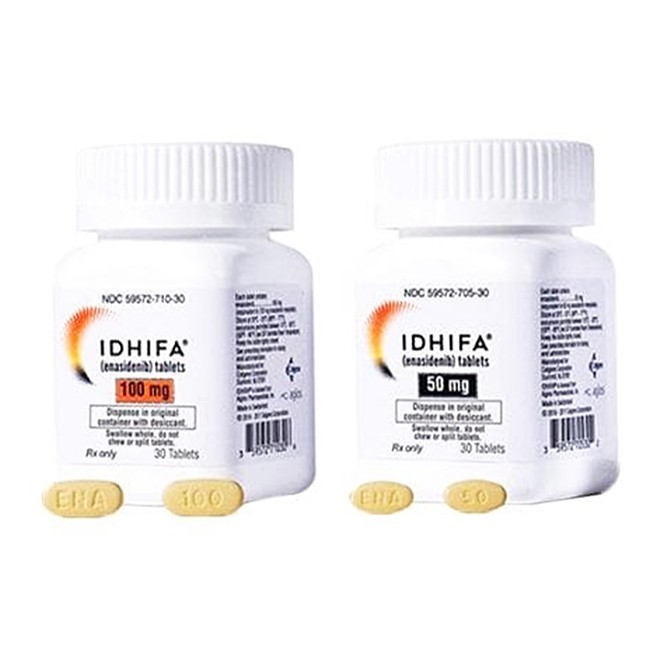
Enasidenib is an isocitrate dehydrogenase-2 (IDH2) inhibitor indicated for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (AML) who harbor an IDH2 mutation.
How to Use Enasidenib
Administration Regimen
Recommended Dosage: 100mg orally once daily, with or without food.
Treatment Course: Continue administration until disease progression or the occurrence of intolerable toxicity. To assess clinical response, treatment for at least 6 months is recommended.
Administration Requirements: Swallow the tablet whole; do not break, crush, or chew it. If a dose is missed or vomiting occurs after administration, take the missed dose as soon as possible on the same day. Resume the regular dosing schedule on the next day, and do not take a double dose to make up for the missed one.
Dosage Adjustment of Enasidenib
Dosage Adjustment for Differentiation Syndrome
Management: If differentiation syndrome is suspected, immediately initiate glucocorticoid therapy (e.g., dexamethasone 10mg every 12 hours) and conduct hemodynamic monitoring.
Dosage Adjustment: If severe pulmonary symptoms requiring intubation/mechanical ventilation or renal dysfunction lasting more than 48 hours occur, discontinue enasidenib temporarily. Resume enasidenib once symptoms improve to grade ≤2.
Dosage Adjustment for Non-Infectious Leukocytosis
Management: Hydroxyurea is the first-line treatment. If hydroxyurea is ineffective, discontinue enasidenib temporarily. Resume enasidenib at the dose of 100mg once daily when the white blood cell (WBC) count decreases to <30×10⁹/L.
Dosage Adjustment for Bilirubin Elevation
If bilirubin levels exceed 3×upper limit of normal (ULN) for ≥2 consecutive weeks without abnormal liver enzymes, reduce the dosage to 50mg once daily.
Once bilirubin levels return to <2×ULN, the dosage may be re-escalated to 100mg once daily.
Enasidenib Use in Special Populations
Pregnant and Lactating Women
Pregnancy Risk: Animal studies have shown that enasidenib has embryofetal toxicity and may cause teratogenicity. Women of childbearing potential must confirm a negative pregnancy test before initiating treatment, and use highly effective contraceptive measures during treatment and for 1 month after the last dose.
Lactation: Breastfeeding is contraindicated during enasidenib treatment and for at least 1 month after the last dose.
Patients with Hepatic or Renal Impairment
Hepatic Impairment: No dosage adjustment is required for patients with mild hepatic impairment (bilirubin ≤ULN and AST >ULN, or bilirubin 1-1.5×ULN). Data on the use of enasidenib in patients with moderate to severe hepatic impairment are insufficient.
Renal Impairment: No dosage adjustment is required for patients with a creatinine clearance (CrCl) ≥30mL/min. Data on the use of enasidenib in patients with more severe renal impairment are limited.